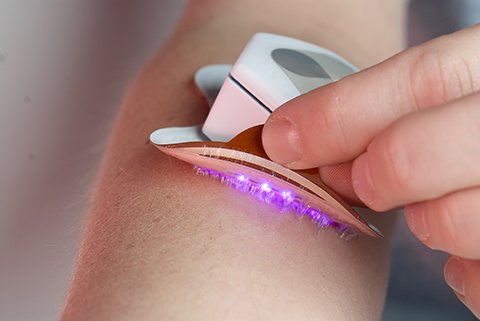
CareWear Corp is a privately held firm based in Nevada that has successfully led through innovation in printed light to advance wearable care for athletes, prosumers and consumers. CareWear has been awarded 66 global patents in addition to receiving international recognition in Optical Engineering (2018 SPIE Startup Challenge Winner), IoT/Wearable Technology (2019 IOT/WT Innovation World Cup® Finalist), and Sports Wearables (2019 WT ISPO Sports Disruptor).
CareWear has developed the first wearable, wireless printed light patches for athletes, prosumers and consumers. CareWear’s light patches are designed to relieve pain, decrease inflammation, accelerate recovery, and improve performance while reducing the likelihood of injury from overuse and muscle fatigue as demonstrated in published research.
CareWear’s medical grade products are FDA registered and available without a prescription, offering a safe, affordable, drug-free therapy solution. Rather than restricting treatments to a clinic or training room, injury and pain relief is now mobile and can be completed during training, at home, or on-the-go in as little as 30 minutes per day. The products are used by professional, collegiate and USA teams, military academies and special forces soldiers.
In the course of this year´s WT | Wearable Technologies Show at MEDICA on Nov 18-21, we have asked CareWear a few questions about their current process of getting CE marking and future steps and goals of the company.
How was the process of getting CE marking for CareWear?
- A CE mark certificate is required to legally market medical devices in the EU. This must comply with the European Medical Devices Regulation (MDR 2017/745).
- Obtaining CE marking has been significantly more challenging as a result of changes to the regulations based on updated requirements in the areas of biocompatibility and clinical performance. This has created a significant backlog and has extended the timelines for approval of new products.
- We have completed all of our audits with our notified body and expect a CE mark certificate by the end of 2019.
What does it mean for you to be CE compliant?
- It means our products have undergone extensive safety testing to the appropriate IEC standards for our Class 2a over-the-counter (OTC) medical device under the MDR, and that the clinical, biocompatibility and safety data provided substantiates the indications for use and labeling and safety requirements.
- It provides the opportunity to commercially market quality medical devices in the EU for safe and effective use by medical practitioners, sports professionals and medical consumers.
What are your next steps and goals for 2020?
- Prosumer marketing in the USA
- Export our products to Canada and Australia where we have established distribution relationships
- Establish new distribution relationships in specific target markets that accept CE or FDA compliance in the professional and consumer OTC markets.
What do you expect from the WT | Wearable Technologies Show at MEDICA?
- CareWear expects to begin sales in the EU and selected global markets in the first quarter of 2020. We plan to establish distribution relationships through contacts we make at WT MEDICA in the professional rehabilitation, sports and consumer markets.
Curious to learn more? Visit CareWear at the WT | Wearable Technologies Show at MEDICA on November 18-21 in Hall 13 Booth D46.