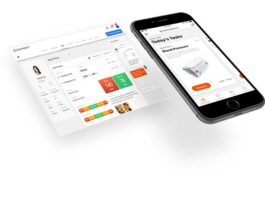
Verisense™ Wearable Sensor Platform for Clinical Trials – Interview with Geoff Gill, president of...
Shimmer, a global leader in wearable technology for research applications, has just launched a next-generation wearable sensor platform. We interviewed Geoff Gill, the president...
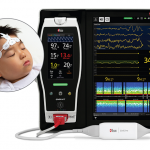
Masimo Receives CE Marking for Next Gen SedLine Brain Function Monitoring for Pediatric Patients
Masimo, an Irvine, California-based manufacturer of noninvasive patient monitoring technologies, received CE marking for its next generation SedLine® brain function monitoring for pediatric patients....
Navitian, iVascular´s New Coronary Microcatheter Receives CE Mark Approval
iVascular, a Barcelona, Spain-based catheter maker, won CE Mark for its new microcatheter called Navitian. The microcatheter can be used to facilitate, guide and...
Janssen’s TREMFYA One-Press Patient-controlled Injector for Adults with Plaque Psoriasis Receives FDA Approval
Janssen Pharmaceutical’s TREMFYA® One-Press, a single-dose, patient-controlled injector for adults with moderate-to-severe plaque psoriasis received FDA approval. TREMFYA is the first FDA-approved medication of...
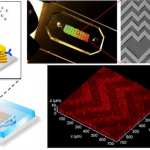
Breakthrough Microfluidic Device Detects Cancer Faster and Less Invasively
Researchers at the University of Kansas, The University of Kansas Cancer Center and KU Medical Center, developed a device that could allow doctors to...
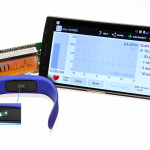
ams Announces new Sensor with Medical-Grade Cardiovascular Monitoring to be Used in Consumer Wearables
ams AG, an Austria-based manufacturer and supplier of sensors worldwide, announced the AS7026, a small optical sensor for continuous cardiovascular health monitoring which delivers...