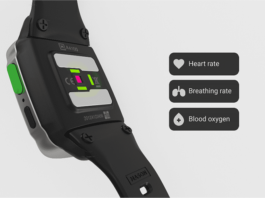
UK Biomedical Company Unveils its geko Wearable Electrostimulation Device at the Orthopedic Summit 2019
Sky Medical Technology, a UK-based biomedical devices company announced that its geko™ device was presented as part of the didactic program as a case...
Profusa Receives CE Mark for Its Wireless Lumee Oxygen Platform
Profusa, a California-based medical device company pioneering the next generation of personalized medicine, announced it has received CE Mark for its Wireless Lumee® Oxygen...
Medtronic Receives CE Mark for its Smart Cobalt and Crome Cardiac Implants
Medtronic announced it has received CE Mark for its Cobalt™ and Crome™ portfolio of implantable cardioverter-defibrillators (ICD) and cardiac resynchronization therapy-defibrillators (CRT-D).
Read more Medtronic...
Denver Startup Receives FDA Clearance for its BioSticker On-Body Sensor for Remote Patient Monitoring
BioIntelliSense, a company making sticker-like sensors that can monitor patient vitals, received FDA 510(k) clearance of the BioSticker™ on-body sensor for scalable remote care....
Remote Patient Monitoring is the Future of CVD Management, Reveals IDTechEx Research
Cardiovascular disease (CVD) is currently the leading cause of death globally. The World Health Organization reported that CVD is responsible for 17 million deaths...
BioHiTech to Provide Its Revolution Series Digesters and Data Analytics Platform to Carnival
BioHiTech Global, a technology and services company that provides cost-effective and sustainable waste management solutions, and Carnival corporation signed a purchase contract where BioHiTech...