Robocath’s Robotic-Assisted Solution R-One Gets CE Mark for Treating Coronary Diseases
Robocath, a Rouen, France-based company that develops robotic systems for treating cardiovascular diseases, won CE certification for its R-One™ robotic device. The CE approval...
Marvell, Samsung Extend Long-Term Partnership for 5G Wireless Networks
Marvell, a leading infrastructure semiconductor solutions provider, expanded it’s a long-term partnership with Samsung for enabling leading wireless infrastructure networks. The two companies are...
STATS’ AI-Powered Technology AutoSTATS Allows Capturing of Sports Tracking Data via Broadcast Video
STATS, a Chicago-based sports data, technology, statistics, and content company, officially launched AutoSTATS, the first artificial intelligence (AI) and computer vision technology to deliver...
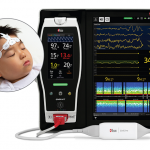
Masimo Receives CE Marking for Next Gen SedLine Brain Function Monitoring for Pediatric Patients
Masimo, an Irvine, California-based manufacturer of noninvasive patient monitoring technologies, received CE marking for its next generation SedLine® brain function monitoring for pediatric patients....
Navitian, iVascular´s New Coronary Microcatheter Receives CE Mark Approval
iVascular, a Barcelona, Spain-based catheter maker, won CE Mark for its new microcatheter called Navitian. The microcatheter can be used to facilitate, guide and...
Janssen’s TREMFYA One-Press Patient-controlled Injector for Adults with Plaque Psoriasis Receives FDA Approval
Janssen Pharmaceutical’s TREMFYA® One-Press, a single-dose, patient-controlled injector for adults with moderate-to-severe plaque psoriasis received FDA approval. TREMFYA is the first FDA-approved medication of...